Why is the top layer of sand crunchy?
Growing up in Florida, two things about the sand stuck out: it’ll burn the shit out of your feet and the top layer has a thin crunchy crust. The first point is pretty easy to explain (it’s Florida), but the second is something a little less obvious.
Every wave that washes up the beach leaves behind a thin film of seawater in the spaces between sand grains. Seawater carries about 35 grams of dissolved salt per liter, mostly sodium chloride. When the sun heats the surface and that water evaporates, the salt has nowhere to go. It stays behind, clinging to the grain surfaces and concentrating in the water that remains.
Water deeper in the sand gets pulled upward through the narrow gaps between grains by capillary action, the same force that draws water up a paper towel. The smaller the spaces, the stronger the pull. Beach sand has pore spaces on the order of tenths of a millimeter, small enough to maintain a steady upward supply of moisture from the water table below. As fast as the surface evaporates, capillary forces are resupplying it with more salt water from underneath. It’s a conveyor belt for dissolved salt, continuously delivering NaCl to the evaporation front at the surface.
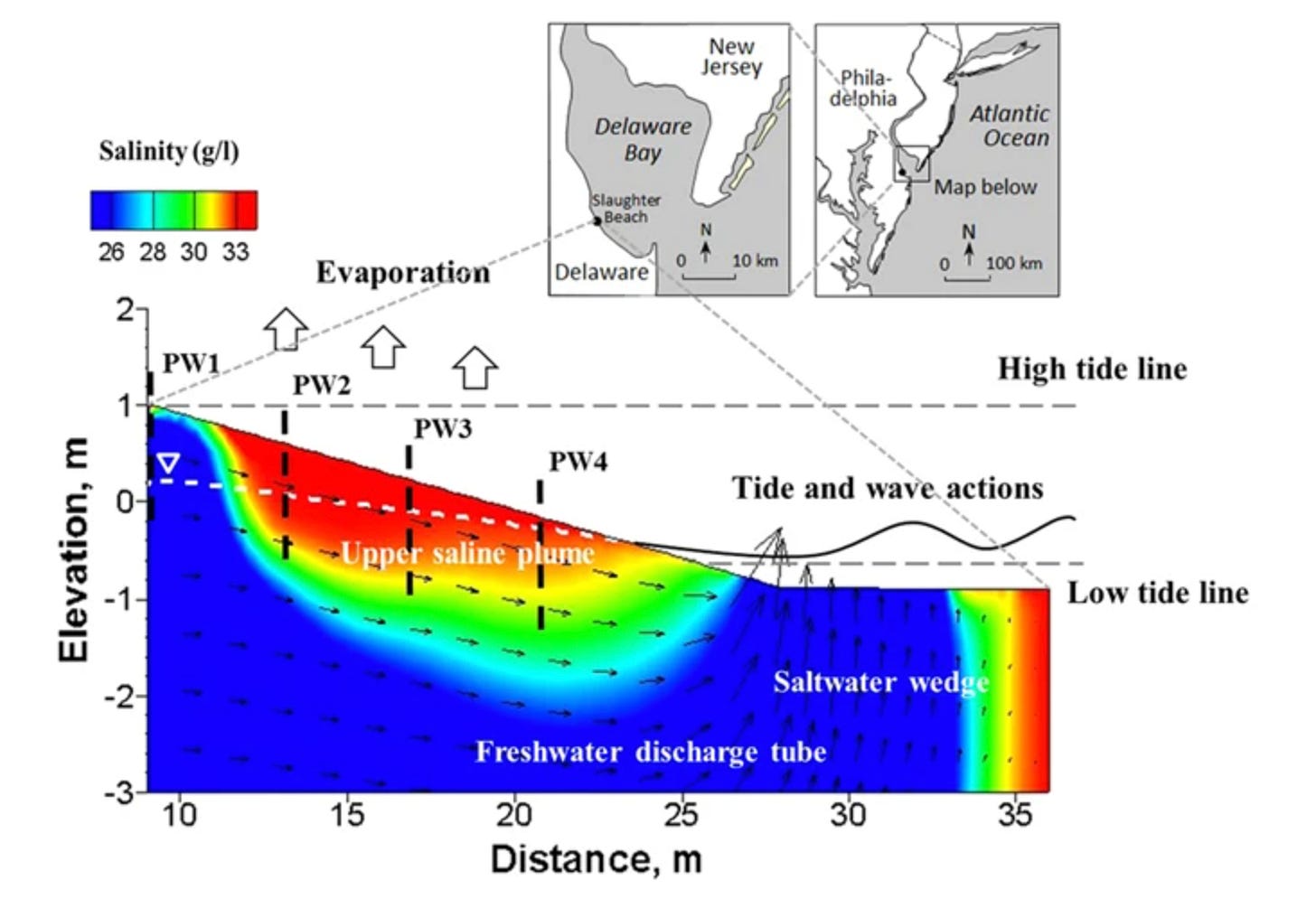
A 2016 study published in Scientific Reports by Geng, Boufadel, and Jackson measured pore-water salinity on an estuarine beach and found that evaporation drove near-surface concentrations to roughly double that of normal seawater. The effect was strongest in the upper intertidal zone during low tide on hot days, when the sand surface was exposed to air and the capillary fringe was actively pumping moisture upward. The deeper sand stayed closer to ambient ocean salinity while extra salt was concentrating right at the top.
Once the pore water at the surface reaches supersaturation, sodium chloride begins to crystallize. The crystals precipitate at the contact points between them, forming tiny mineral bridges that lock neighboring grains together. The same process destroys ancient stone buildings and monuments over centuries, salt crystals wedging into pore spaces and generating enough pressure to crack rock from the inside. On a beach, the result is less dramatic but mechanically identical: a thin brittle pancake of sand held together by crystalline NaCl cement, just waiting to be tossed and shattered.
The crust has almost no flexibility. Salt crystals are rigid and the connections between grains are thin. One footprint is enough to collapse the structure back into loose sand. Underneath, where evaporation hasn’t concentrated the salt, the grains never bonded in the first place.
The crust forms best on hot, calm days with low humidity, above the high tide line where the sand hasn’t been recently swashed. Fog or a high tide dissolves the crystals and resets the process and morning dew can weaken it. You’ll rarely feel the crunch on a beach that’s been washed by waves in the last few hours, but give the upper beach a full day of sun and it’ll be there.
Further Reading:


Great piece again Kevin. You should try surfing in the UK - the sand here is coooold! It’s actually quite good for your health - the cold sand has an earthing effect, neutralising the the ions in your body, which leads to enhanced circulation, reduced inflammation, and boosted immunity 🤙